-
If you are citizen of an European Union member nation, you may not use this service unless you are at least 16 years old.
-
You already know Dokkio is an AI-powered assistant to organize & manage your digital files & messages. Very soon, Dokkio will support Outlook as well as One Drive. Check it out today!
| |
Relating Kinetics to Thermodynamics
Page history
last edited
by Mike Gysin 10 years, 11 months ago
Relating Kinetics to Thermodynamics
Introduction:
Thermodynamics is the study of heat and how it relates or can be converted to other forms of energy and work. Thermodynamics tell us about a system and how that system would change if it were to exist in different states. Kinetics considers the rates of chemical reactions and processes. It takes into consideration what conditions influence the rate of the reaction and what the yield of the reaction may be. Kinetics also provides information about the mechanism of the reaction and possible transition states. The law of mass action is an important part of chemical kinetics, it states that the speed of a chemical reaction is proportional to the quantity of the reactants. The study of thermodynamics can tell us about which processes can happen, but not necessarily which processes will happen. To investigate this, we must relate our study of thermodynamics to kinetics.
The Basics:
-
There are two different forms of ∆G involved in thermodynamics and in kinetics. ∆Gr measures the difference in Gibbs energy between the reactants and products. ∆Ga, also known as Ea, measures the difference between reactants and the transition state, which is the state of highest energy that the reactants go into before reaching the products. Ea appears in the Arrhenius equation.
-
The Arrhenius equation is an equation that relates the rate constant k to the activation energy.

where k=rate constant, A=Arrhenius factor, Ea=activation energy for that step of the reaction, R=universal gas constant (given as 8.314 J/(mol*K)), and T=temperature.
-
The Arrhenius equation doesn’t tell us the actual rate of the reaction; it only tells us the rate constant for that step of the reaction.
-
A negative ∆G does not necessarily mean a large k value. k depends on Ea only. ∆G only tells the direction of the reaction, not the speed.
-
∆G is a state function, therefore it only depends on the initial state of the reaction (reactants) and the final state of the reaction (products).
In contrast Ea affects the rate of reaction and can be decreased with a catalyst, such as an enzyme, which helps to stabilize the transition state and lower the Gibbs energy of the step. The activation energy is the “energy barrier” which is the energy required to reach the transition state. In fact, activation energy can be used to determine which step in a multistep reaction is the rate limiting step. The step with the highest activation energy is always the rate limiting step, which is the slowest step in the reaction. The transition state is the top of the hill on the graph.

Reaction Mechanisms:
-
The reaction mechanism is the sequence of elementary steps that describe the overall reaction. The mechanism includes possible intermediates, activated complexes, and transition states. The reaction mechanism also provides valuable information as to which bonds are broken and which bonds are formed as a result. For example, the reaction mechanism of the process A + B -> C, as seen below, shows an intermediate D that is produced and then consumed.

Law of Mass Action:

-
Keq is not the same thing as k, the rate constant. The equilibrium constant tells the final composition, at equilibrium. The rate constant tells the speed of the reaction. This can also be seen in the units of measurement (no unit v [molarity]/s). K is also directly related to ∆G of the reaction, which determines which reaction are favorable. If reactions are not at equilibrium, then we can calculate Q, the mass action ratio, which will determine if the reaction is favorable. If Q is greater than K, then the reaction is unfavorable because there are more products than expected at equilibrium, which means the reverse reaction will be favored, turning products into reactants to reestablish equilibrium. If Q is less than K, then the reaction is favorable, following similar reasoning as above. Q is found by the same formula as K, except the value is not equal to the equilibrium constant.
What does it mean that a reaction can be thermodynamically favorable, but kinetically unfavorable?
-
A thermodynamically favorable reaction means that the total Gibbs energy of the products is lower than that of the reactants. Basically, the entropy of the universe increases, which means that it is a spontaneous process by the second law of thermodynamics.
-
A kinetically unfavorable reaction depends more on practicality than chemical limitations. Any thermodynamically favored reaction will progress toward equilibrium. Some reactions, however, go so slow that there is no noticeable change in the reaction mixture, even though ∆G is negative. Therefore, a reaction that goes at such a slow rate that it does not occur in a reasonable amount of time is kinetically unfavorable (such as diamond to graphite, which would take millions of years).
-
A kinetically unfavorable reaction has a very high activation energy. This is opposed to a kinetically favorable reaction, which has a relatively low activation energy. A thermodynamically unfavorable reaction would have a positive ∆G.
|
|
Thermodynamically Favorable
|
Thermodynamically Unfavorable
|
Kinetically Favorable
|
Kinetically Unfavorable
|
|
Information
|
Notifies you on which direction is favored
|
Notifies you on how fast the reaction will go
|
|
Products or Reactants
|
Products are favored
|
Reactants are favored
|
-
|
-
|
|
Rate
|
-
|
-
|
Faster
|
Slower
|
|
How you determine?
|
If K is greater than Q
|
If K is less than Q
|
Using the Equation
|
|
What is K?
|
K is the equilibrium constant with the ratio of products to reactants at equilibrium
|
K is the rate of an elementary step within the reaction mechanism.
|
What happens when a reaction can follow two different reaction pathways? Explain the difference between kinetic reaction control and thermodynamic reaction control. What are some examples of these types of reactions? Are they common to chemistry?
-
A reaction can be under two different kinds of control, which results in two different pathways. These pathways have different activation energies, meaning they have different rates of reaction. When a reaction is under kinetic reaction control, the main product will be the product that forms faster. Thermodynamic reaction control favors the product that releases more Gibbs energy. The pathway under thermodynamic control generally has a higher activation energy, meaning it forms slower. These reactions generally are increased through an increase in heat. Since there are multiple pathways, the reaction will product a mixture of products, generally isomers. Many reactions follow this path, most famously the Diels-Alder set of reactions. The products of Diels-Alder reactions are generally a set of isomers, and are very common in biosynthetic processes. Two possible isomers are formed in a Diels-Alder reaction. The ratio of these isomers are dependent on the conditions under which the reaction is placed. In a higher temperature, since all molecules will have the energy used to overcome the activation energy, the thermodynamically favored product is formed. Otherwise, the kinetically favored product is formed.
Reaction Rate:
-
Reaction rate is the speed at which the reactant’s and product’s concentrations change. This rate of change is useful to describe the rate of the reaction. It is useful to view this in a time v concentration graph, where the concentration of a product and a reactant is shown to be changing as time increases. The reaction shown below is the conversion of one mol of hydrogen gas and one mol of iodine (shown in red) to 2 mol of hydrogen iodide (shown in blue). As the amount of reactant decreases, there is an equal and opposite increase in products, as seen below.
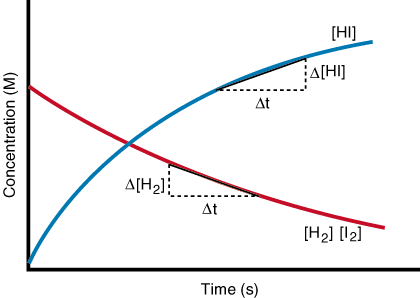
How can heating a system affect the reaction rate? What equations predict that heating
the system will change the reaction rate?
-
The reaction rate is a measure of how fast or slow a reaction takes place. For a generalized reaction aA + bB → pP + qQ, the reaction rate is given by, where the derivative indicates the change in concentration at a moment in time:

-
When heat is added to a system, the particles in the system will be moving at faster average speeds and so, will have more frequent collisions. According to collision theory, particles can only react when they collide, so a greater frequency of collision will result in an increase in reaction rate. Moreover, not all collisions will result in the reaction taking place; collisions between particles must have enough energy to get the reaction started. This minimum amount of energy required to start the reaction is called the activation energy. So, as heat is added and particles move with greater speed, more particles will also collide with enough energy to result in the reaction taking place. Thus, adding heat to a system will increase the reaction rate.
-
An equation that shows the temperature dependence of the rate constant is the Arrhenius equation:

As temperature increases, the exponential term gets larger and thus, the rate constant also gets larger, leading to an increase in the reaction rate.
How can a catalyst affect the reaction rate? What quantity does the catalyst affect?
(which ∆G/Ea is affected?) What are some examples of catalysts, and how are they used
in biochemistry?
-
A catalyst is a substance that increases the reaction rate without being consumed in the reaction itself. The presence of a catalyst affects the quantity of activation energy needed, lowering this energy barrier so that more molecular collisions will have the minimum amount of energy needed for the reaction to proceed. With a greater number of molecular collisions resulting in product, the reaction rate is increased since Ea, not ∆G, is changed.
-
Catalysts work as to stabilize the activation energy of the reaction. For example, they could stabilize a protonated intermediate or a radical through interactions with the molecules of the catalyst - in enzymes, interactions with amino acid side chains serve as means to change the pH of a solution to promote protonation or deprotonation, which results in a reaction.
-
Catalysts are commonly used in science and biochemistry; almost all reactions that occur in living cells are driven by biological catalysts called enzymes. They allow reactions to take place at rates that are practical to support life. An example of a catalyst is iron catalyzing the formation of ammonia from nitrogen gas and hydrogen gas or hexokinase phosphorylating glucose in glycolysis.
Common Pitfalls:
Some students confuse big K (equilibrium constant) and little k (rate constant). Explain the differences between these two properties.
In the generalized reaction aA+bB--> pP+qQ, the equilibrium constant is defined as:

From this, we see that if K>1, this reaction will favor the products, and if K<1 it will favor the reactants. Therefore K tell us the ratio of products to reactants.
-
The rate constant, k, tells us the rate of an elementary step in the reaction mechanism. k can be found experimentally, or by solving the Arrhenius equation discussed above. In that case we get:

-
The equilibrium constant, K, is used in thermodynamic problems, while the rate constant, k, is used in kinetics problems.
-
For example, for any reaction, the equilibrium constant can be calculated from a ratio of the concentration of products to reactants. Then, in order to determine the direction a reaction will proceed for any combination of the reactants and products, a reaction quotient, Q, can be calculated and compared to the value of K. However, this information would not tell us anything about the rate constant, k. In contrast, to consider the rate constant, if there is a large activation energy, then the rate constant will be small, but this would not affect the equilibrium constant.
Questions:
-
True or False. A larger ∆Gr means that the rate constant k is increased as well.
-
Which reaction conditions would go to equilibrium first?
-
∆G = -20 kJ/mol, k = 0.37 [molarity]/s
-
∆G = -100 kJ/mol, k = 0.04 [molarity]/s
-
∆G = 32 kJ/mol, k = 1.27 [molarity]/s
Works Cited:
http://en.wikipedia.org/wiki/Kinetics
http://www.science.uwaterloo.ca/~cchieh/cact/c123/massacti.html
http://web.tock.com/kalee/chem32/kinT/
http://en.wikipedia.org/wiki/Reaction_mechanism
http://www.chm.davidson.edu/vce/kinetics/ReactionRates.html
http://www.google.cm/imgres?q=concentration+time+graph&um=1&hl=fr&biw=922&bih=914&tbm=isch&tbnid=tlLQvLuJAZXd8M:&imgrefurl=http://www.sparknotes.com/chemistry/kinetics/ratelaws/section1.rhtml&docid=CqU2hJUR8tta6M&imgurl=http://img.sparknotes.com/figures/D/db7818cd37ba1ff8b6734f17e9b15a18/graph.gif&w=420&h=298&ei=1oihULbZMcq-2gWF14DwBg&zoom=1&iact=hc&vpx=339&vpy=157&dur=1458&hovh=189&hovw=267&tx=112&ty=113&sig=117357371955168269859&page=1&tbnh=144&tbnw=203&start=0&ndsp=19&ved=1t:429,r:1,s:0,i:66
http://www.chemguide.co.uk/physical/basicrates/temperature.html
http://www.google.com/imgres?um=1&hl=en&sa=N&tbo=d&biw=922&bih=914&tbm=isch&tbnid=TvMT7eSjCfqxRM:&imgrefurl=http://redslime.typepad.com/h_ap_biology/unit-1-ap-thermodynamics-enzyme-function.html&docid=b7lUFkyYj8D59M&imgurl=http://redslime.typepad.com/.a/6a00e551f28d3c88340120a4d22158970b-320wi&w=320&h=243&ei=8omhUPvwAuiM2gWmn4DgCw&zoom=1&iact=rc&dur=163&sig=103711666789497921244&page=1&tbnh=133&tbnw=176&start=0&ndsp=23&ved=1t:429,r:10,s:0,i:104&tx=118&ty=111http://en.wikipedia.org/wiki/Activation_energy
http://web.tock.com/kalee/chem32/kinT/#2
Problem Set Answers:
1. False. k does not depend on ∆Gr.
2. i. Even though it is the less favorable reaction thermodynamically when compared to ii., it has a higher rate constant. iii. will never go to equilibrium in that direction because ∆G is positive.
Reaction Rate
How can heating a system affect the reaction rate? What equations predict that heating
the system will change the reaction rate?
Relating Kinetics to Thermodynamics
|
|
Tip: To turn text into a link, highlight the text, then click on a page or file from the list above.
|
|
|
|
|
Comments (0)
You don't have permission to comment on this page.